Our rate of effusion calculator is based on this formula and can be used to calculate either rate of diffusion/effusion or molar masses of gases, whichever suits your need. 
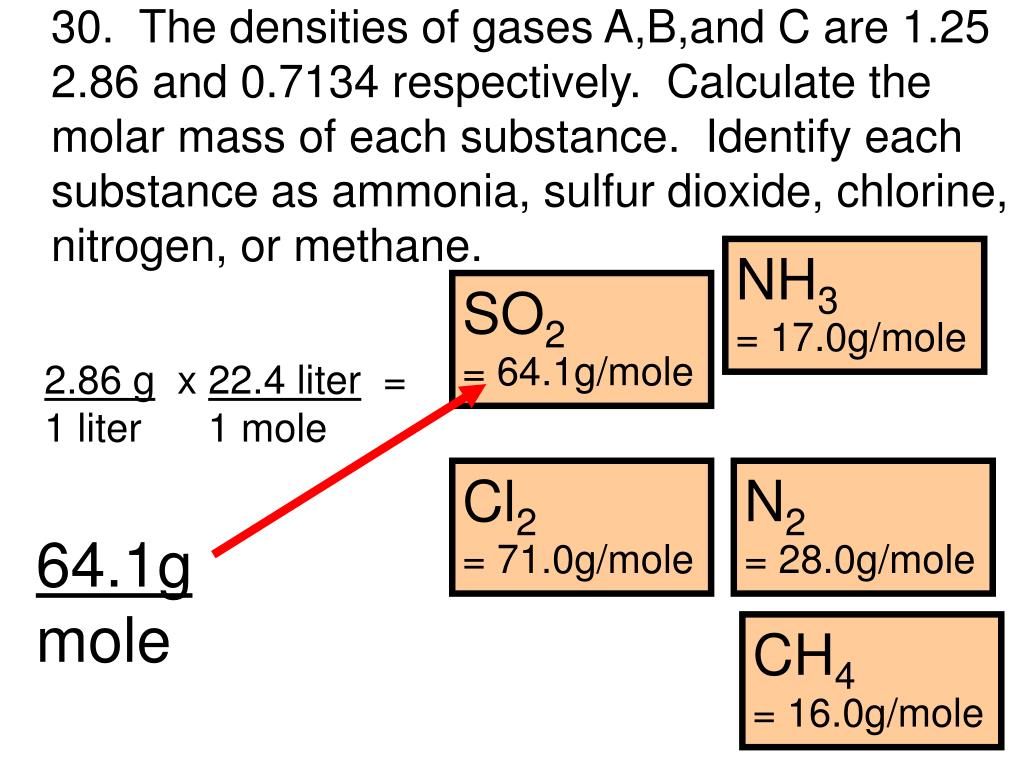
M 1 \text M_1 M 1 and M 2 \text M_2 M 2 - Molar masses of gases.v 1 \text v_1 v 1 and v 2 \text v_2 v 2 - Rates of diffusion or effusion and.The molar mass of NH3 (ammonia) is 17.031 g per mole. This is because hydrogen chloride has almost twice the molecular weight of ammonia. Rate 1 / rate 2 = mass 2 / mass 1 \text v 1 / v 2 = M 2 / M 1 Ammonia is a chemical compound that has the formula NH3, being made out of one nitrogen atom and three hydrogen atoms. Urea is formed by the reaction of ammonia gas and carbon dioxide gas. 6 NH 3 (molar mass 267.5 g mol1) is passed through a cation exchanger. 
You can write the formula for Graham's law of diffusion or effusion of gases as: Molar mass of ammonia ( NH 3) ( Atomic mass of nitrogen) x 1 ( Atomic mass of hydrogen) x 3 ( 14 x 1) ( 1 x 3) 17 g Molar mass of ammonia (NH 3) is 17 g. 2009 (a) Co(en) (NH3 ) 2 2 (b) Co(H 2O) 4 (en)3 (c) Co(en) 2 (NH3 )2. a) (1 pts) Calculate the mass percent of Cl2. The rate of diffusion or effusion of a gas is inversely proportional to the square root of its molecular weight. Molar mass of Ni2 , NH3 and Cl2 are 58.69 g/mol, 17.04 g/mol, and 70.90 g/mol, respectively. Graham's law of diffusion definition for gases states that: One mole of Dinitrogen N2 and three moles of Dihydrogen H2 react to form two moles of Ammonia NH3 Show Chemical Structure Image Reaction Type Synthesis Redox Reversible reaction (equilibrium) Redox (Oxidation-Reduction) Reaction N2 H2 NH3 might be a redox reaction. The diffusion of gases refers to the process in which the molecules of one gas distribute themselves into another gas. Ammonia NH3 or H3N CID 222 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. Diffusion, in general, refers to the process in which the particles of a substance move from an area of higher concentration to an area of lower concentration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
